Complete Material Lifecycle
GMP-Compliant Tracking
End-to-end material management from receipt to destruction. Track material lots, inventory, allocation, consumption, retention, and destruction with complete GMP compliance and real-time dashboards for threshold monitoring and reordering.
Key Material Management Capabilities
Comprehensive features for complete material lifecycle management
Lot Tracking
Complete lot-based tracking with unique identifiers and full traceability
Auto Consumption
Automatic consumption tracking during procedure execution
FEFO Rotation
First Expire, First Out stock rotation automatically enforced
Threshold Alerts
Multi-level alerts for materials approaching reorder points
GMP Compliance
21 CFR Part 11 compliant with complete audit trails
Destruction Workflows
Complete destruction workflows with certificates and chain of custody
Analytics
Consumption analytics and inventory optimization insights
Full Traceability
Forward and backward traceability for all materials
Complete Material Lifecycle
From receipt to destruction—every stage tracked with GMP compliance and complete traceability
Receipt
Material receipt with supplier lot verification
- Supplier lot number assignment
- Identity verification
- Quarantine status
- Labeling and documentation
Testing & Approval
Quality testing and status release
- Quality testing workflow
- Status change to Approved
- Specification verification
- Electronic signature capture
Storage
Controlled storage with environmental monitoring
- Controlled environment tracking
- Status-based segregation
- FEFO stock rotation
- Expiry date monitoring
Allocation
Material allocation to procedures
- Procedure-based allocation
- Lot selection (FEFO)
- Reservation and availability
- Allocation confirmation
Consumption
Usage tracking and inventory deduction
- Automatic consumption tracking
- Real-time inventory updates
- Usage documentation
- Return workflows
Retention/Destruction
Retention sample or destruction
- Retention sample management
- Destruction workflows
- Chain of custody
- Certificate generation
GMP Compliance Built-In
Full regulatory compliance with FDA, EMA, and WHO requirements for pharmaceutical material management
Complete Audit Trails
Every material transaction logged with user identity, timestamp, reason codes, and electronic signatures. Full forward and backward traceability.
Regulatory Documentation
Comprehensive documentation for FDA, EMA, and WHO compliance. Retention periods, batch records, and testing documentation maintained per regulations.
Status Workflow Automation
Automated status transitions from Quarantine to Approved upon testing completion. Automatic status changes for expired materials with alerts.
Segregation & Control
Physical and system-based segregation by status and material type. Controlled access, dual authorization for critical operations, and environmental monitoring.
Regulatory Compliance Standards
Material Management Features
Comprehensive capabilities for complete material lifecycle management with GMP compliance
Material Lots & Inventory Tracking
Complete lot-based tracking with unique identifiers for every material. Full forward and backward traceability from receipt to final disposition.
- Unique lot/batch number assignment at receipt
- Complete lot lineage tracking (supplier lot → internal lot → usage)
- Real-time inventory calculations across all lots
- Lot status management (Quarantine, Approved, Rejected, Expired)
- FEFO (First Expire, First Out) stock rotation
- Multi-location inventory tracking
- Lot splitting and merging capabilities
- Complete lot history and audit trail
Inventory Allocation & Consumption
Automated allocation and consumption tracking during procedure execution. Real-time inventory deduction with complete usage documentation.
- Automatic material allocation during procedure runs
- Real-time consumption tracking and inventory deduction
- Material reservation and availability checking
- Lot selection based on FEFO and availability
- Partial consumption and return workflows
- Usage linked to specific procedures and samples
- Consumption history and reporting
- Automatic inventory reconciliation
Retention & Destruction Management
Complete GMP-compliant retention and destruction workflows. Track material retention samples, destruction certificates, and chain of custody.
- Retention sample management and tracking
- Destruction workflow with approval routing
- Chain of custody documentation
- Destruction certificates and witness signatures
- DEA Form 41 support for controlled substances
- Non-retrievable destruction verification
- Retention period tracking and alerts
- Complete destruction audit trail

Material Dashboards & Analytics
Real-time material dashboards with threshold monitoring, reorder alerts, and comprehensive analytics for inventory optimization.
- Real-time inventory status dashboard
- Threshold limit monitoring and alerts
- Materials at critical/warning levels
- Ready-to-reorder recommendations
- Consumption rate analytics
- Expiry date tracking and alerts
- Inventory trend analysis
- Customizable dashboard views
GMP Compliance & Traceability
Full GMP compliance with complete audit trails, electronic signatures, and regulatory documentation for all material transactions.
- 21 CFR Part 11 compliant electronic signatures
- Complete audit trail for all material transactions
- ALCOA+ data integrity principles
- Status workflow automation (Quarantine → Approved)
- Supplier verification and approval tracking
- Environmental condition monitoring
- Segregation by status and material type
- 7-year record retention compliance
Threshold Limits & Reordering
Automated threshold monitoring with intelligent reorder recommendations. Prevent stockouts and optimize inventory levels.
- Configurable threshold limits per material
- Multi-level alerts (Normal, Warning, Critical)
- Automated notification system
- Reorder point calculations
- Consumption-based forecasting
- Supplier lead time considerations
- Bulk reorder recommendations
- Email and in-app notifications
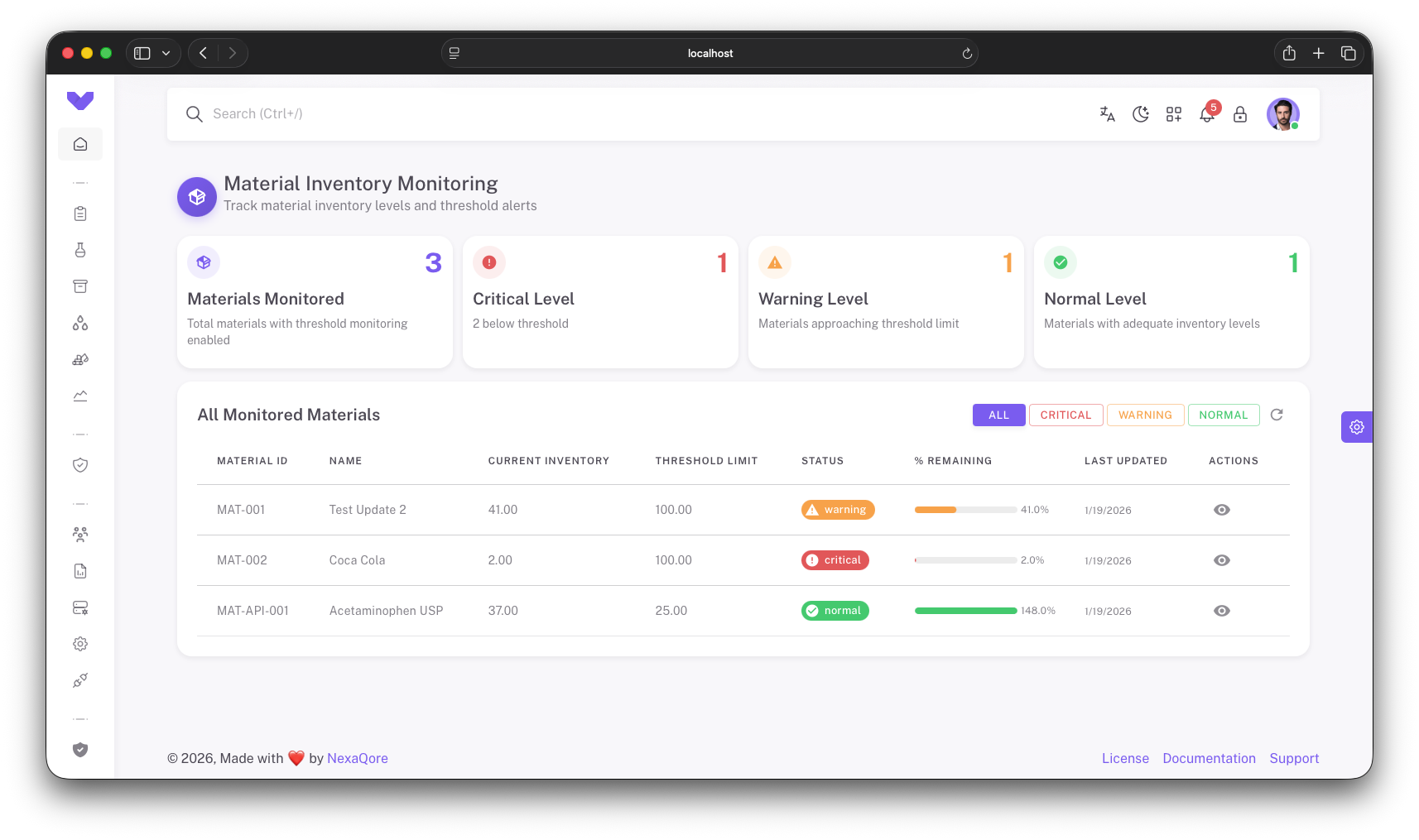
Real-Time Material Dashboards
Monitor inventory levels, threshold alerts, and reorder recommendations with comprehensive analytics
Inventory Status Dashboard
Real-time view of all materials with status indicators, current inventory levels, and threshold status
Threshold Monitoring
Automated alerts for materials at warning (≤50% threshold) and critical (≤20% threshold) levels
Reorder Recommendations
Intelligent reorder suggestions based on consumption rates, lead times, and current inventory levels
Consumption Analytics
Track consumption trends, identify patterns, and optimize inventory levels based on historical data
Complete Transaction Tracking
Track every material transaction with complete lot history, consumption, returns, and destruction records
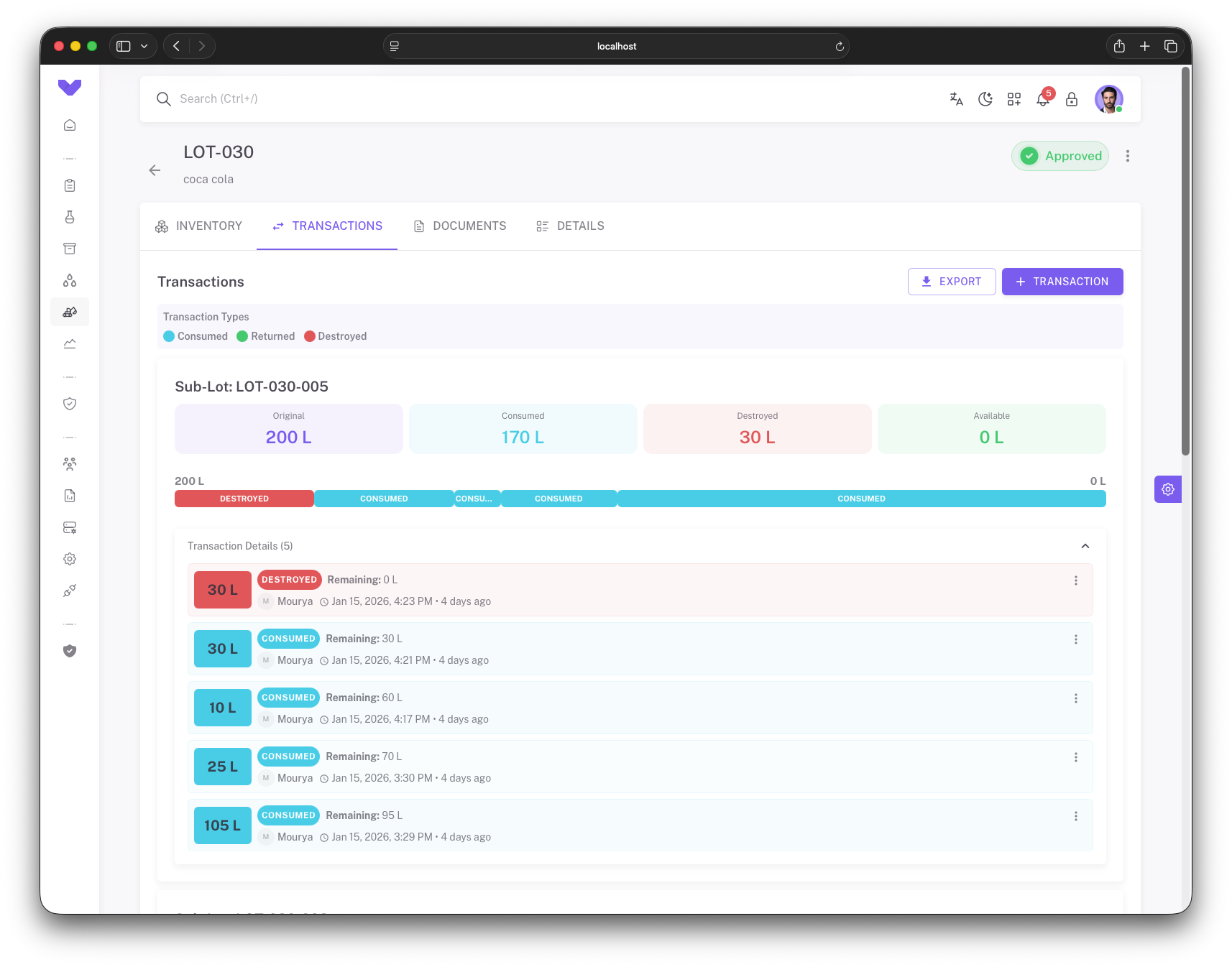
Transaction History
Complete audit trail of all material transactions including consumption, returns, and destruction with timestamps and user information
Sub-Lot Tracking
Track material allocation across sub-lots with visual progress bars showing consumed, returned, and destroyed quantities
Transaction Details
Detailed transaction records with quantity, type, remaining inventory, user attribution, and relative timestamps
Status Management
Real-time status tracking with approved states, transaction type filtering (Consumed, Returned, Destroyed), and export capabilities
Ready to Transform Your Material Management?
Experience complete GMP-compliant material lifecycle management. Track every material from receipt to destruction with real-time dashboards and automated workflows.